Key findings
- Durable effect observed ≥7 years from first infusion
- Sustained preservation of endogenous insulin production at group level
- Clear dose-dependent trends over long-term follow-up
NextCell Pharma AB announced long-term follow-up results from its ProTrans-Repeat study, reporting sustained preservation of endogenous insulin production in patients with newly diagnosed type 1 diabetes. ProTrans is NexCell’s umbilical cord-derived mesenchymal stem cell therapy candidate.
According to the company, the new analysis extends to approximately 6 years after the last ProTrans infusion, which is roughly 7 to 7.5 years since the first infusion.


At this latest follow-up, NextCell has found different trends by dose group:
- High-dose: Preserved endogenous insulin production, and two of three patients continued to show insulin production close to baseline, while one showed a more pronounced decline.
- Intermediate-dose: Limited preservation.
- Low-dose: Progressive decline over time.
NextCell argues that the preservation of insulin production for more than seven years after the first infusion is unexpected in type 1 diabetes, and that the dose-dependent pattern supports the biological rationale for ProTrans.
“A single infusion of ProTrans has demonstrated such a high and durable clinical impact that our strategy is to bring this treatment to patients as quickly as possible. At the same time, I am encouraged by the long-term results from ProTrans-Repeat, which show sustained efficacy over extended follow-up and confirm that ProTrans can also be safely and effectively administered repeatedly if needed,” says Mathias Svahn, CEO of NextCell.
What’s next?
NextCell says its primary objective is to pursue ProTrans as a single-infusion treatment and seek market approval on that basis, since that is where they have the most data. Following market approval, NextCell intends to expand the label to include pediatric patients and evaluate repeated ProTrans infusions to potentially maximize and prolong the effect in selected patient groups.
Here’s their current pipeline:
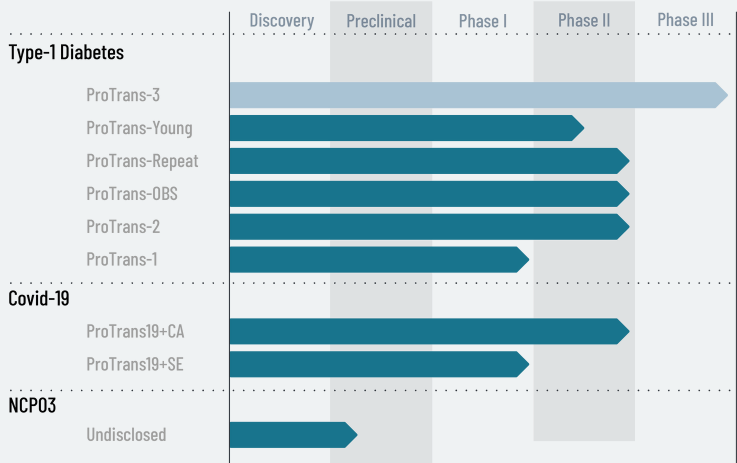
Company snapshot
NextCell Pharma is a clinical-stage cell therapy company developing ProTrans, a patent-protected platform based on allogeneic mesenchymal stromal cells (MSCs) derived from umbilical cord tissue. NextCell uses a proprietary selection algorithm to tailor the cells to specific indications. The company states that a Phase III trial is planned to begin after it secures a commercial partner. ProTrans is also being evaluated for other autoimmune and inflammatory conditions.
NextCell’s subsidiaries include Cellaviva (a private stem cell bank) and QVance (a provider of quality services for developers of advanced therapies).
Want to keep up on regenerative medicine? Get the weekly newsletter here.


















