Cellino and Matricelf announced a collaboration to advance the biomanufacturing of personalized spinal cord injury treatments.
According to the companies, integrating Cellino’s automated induced pluripotent stem cell (iPSC) manufacturing system with Matricelf’s double autologous 3D differentiation process may lead to scalable, patient-specific regenerative therapies.
Nebula™, Cellino’s proprietary closed-cassette biomanufacturing system, is designed for consistent and sterile production of induced pluripotent stem cells (iPSCs) at scale. The company reports that this innovation enables on-demand and contamination-free iPSC production, bringing regenerative medicine closer to practical clinical applications.


As part of this collaboration, Cellino has successfully manufactured and provided autologous iPSC lines (stem cells derived directly from each patient’s own cells) from four different donors to Matricelf, a biotech company focused on regenerative therapies for spinal cord injuries.
Matricelf’s technology incorporates an extracellular matrix-based hydrogel produced from the patient’s own omentum (a fatty tissue layer in the abdomen), which facilitates precise cell differentiation and tissue formation without the need for immunosuppressive drugs.
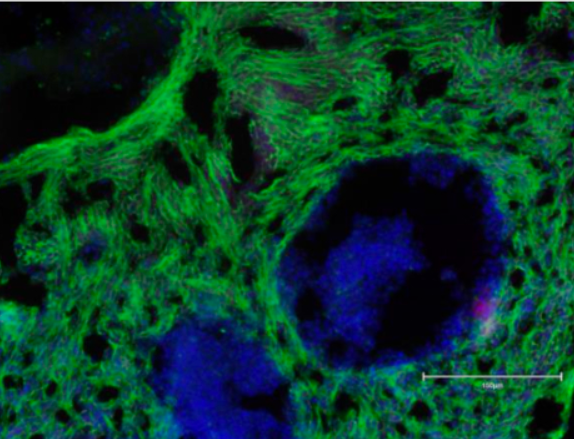
Matricelf reports that it has successfully differentiated the Cellino-generated iPSCs into functional neural tissues exhibiting synchronized electrical activity, a key indicator of functional neural networks. Rigorous industry-standard assays confirmed crucial neural characteristics including genetic and protein marker expression. Matricelf states that these results from the engineered tissues produced with Cellino’s iPSCs were comparable to those previously obtained using Matricelf’s own iPSC-derived tissues.
With the aim of developing new treatment strategies for spinal cord injury, Matricelf plans to submit an Investigational New Drug (IND) application next year to initiate clinical trials of its autologous iPSC-derived therapy. This approach addresses a significant unmet medical need and has the potential to restore function in individuals affected by spinal cord injuries.
“Collaboration on this international scale is a vital step toward our vision of a fully autologous regenerative therapy for spinal cord injury,” said Gil Hakim, CEO of Matricelf. “Matricelf’s innovative platform, in combination with Cellino’s Nebula™ technology, is setting new standards in scalable patient-specific tissue engineering, bringing us closer to real-world applications of regenerative medicine.”
“The ability to produce high-quality, patient-specific iPSCs at scale is poised to revolutionize regenerative medicine,” said Marinna Madrid, Ph.D., Co-Founder and Chief Product Officer, Cellino. “Our collaboration with Matricelf exemplifies how international collaboration can accelerate innovation and expand access to life-changing therapies.”
The collaboration underscores the commitment from both companies to leverage advanced technologies—including AI-driven automation, high-throughput biomanufacturing, and sophisticated neural tissue engineering—to accelerate the development and commercialization of regenerative therapies for neurological diseases.
About Cellino
Cellino develops advanced biomanufacturing technology aimed at enabling personalized cell, tissue, and organ replacements. More information can be found at cellinobio.com.