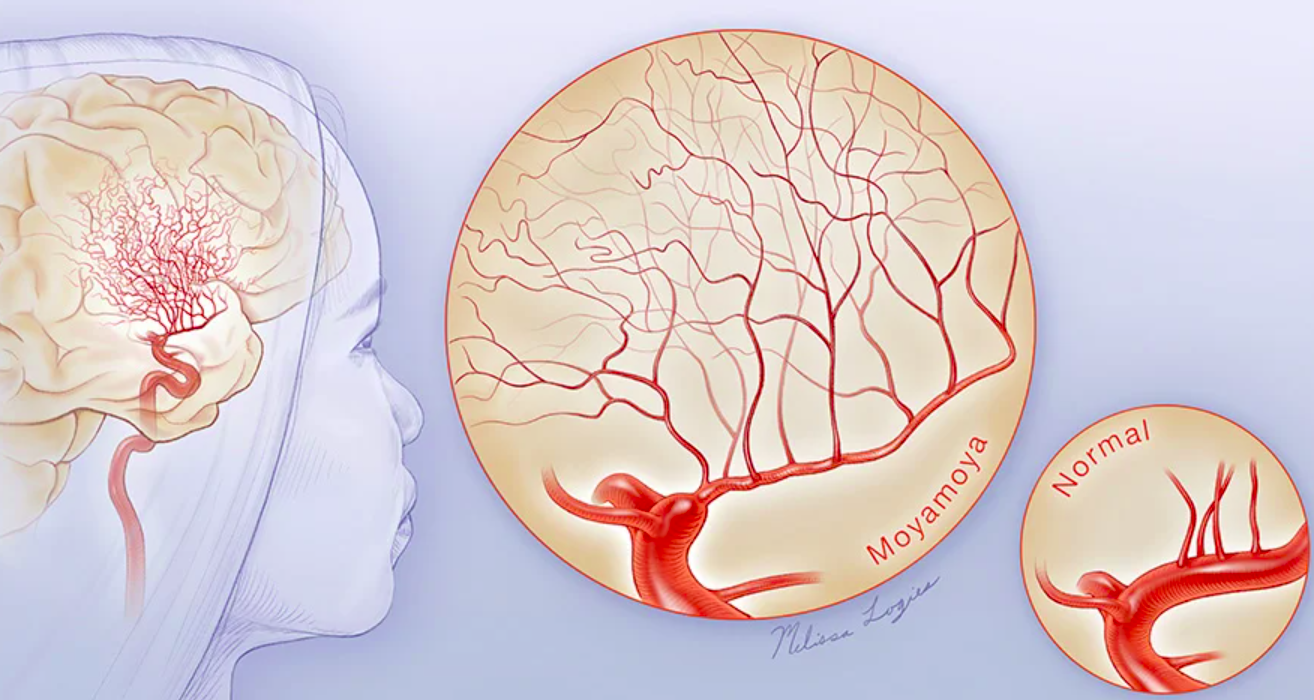
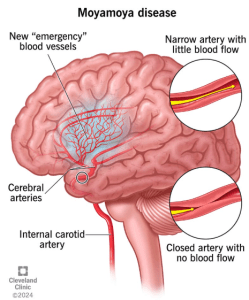
Moyamoya disease is a chronic cerebrovascular disorder characterized by progressive narrowing and occlusion of arteries at the base of the brain, leading to the development of fragile, abnormal vascular networks. This results in an increased risk of strokes, both ischemic and hemorrhagic. Current surgical approaches aim to restore blood flow, but not all patients benefit, and innovative therapies are being sought to improve outcomes.


(Image – Cleveland Clinic)
In this single-arm, open-label trial, up to nine adult patients (ages 35-65) with persistent cerebral ischemia after temporal muscle flap surgery will receive local transplantation of iPSC-EVs, supplied by iRegene Therapeutics Co., Ltd. The study design includes:
- Administration of iPSC-EVs at the surgical site to potentially enhance angiogenesis and collateral circulation formation.
- Monitoring and assessment at multiple time points: 3 days, 2 weeks, and 1 month post-treatment for primary outcomes, with extended follow-up to 24 months for secondary outcomes.
Researchers will primarily evaluate:
- Safety and tolerability: Tracking adverse events and treatment-emergent effects in the short term.
- Imaging outcomes: MRI and transcranial Doppler (TCD) before and at 1 month after iPSC-EV treatment to assess changes in cerebral blood flow.
- Neuropsychological function: Standardized tests (mRS, MMSE, MoCA) before and at 1 month to evaluate cognitive and functional status.
Secondary outcomes include longer-term follow-up with additional imaging (DSA, CT, HR-VWI) and neuropsychological assessments at 3, 6, 12, and 24 months to track durability and function of reconstructed blood flow.
According to the researchers, iPSC-derived extracellular vesicles may offer several potential advantages:
- Promoting vascular regeneration and enhancing collateral blood vessel formation.
- Suppressing inflammatory responses in the brain.
- Stable, drug-like production and potential for future genetic engineering.
The team notes that iPSC technologies are gaining attention worldwide for their regenerative and tissue-targeting capacities. Exosome-based therapies, in particular, are being explored for their ability to deliver bioactive molecules and influence cell repair processes.
The trial began in May 2025 and is expected to complete primary outcome collection by May 2026, with overall completion projected for October 2026. Once finished, investigators will report on safety, neuroimaging changes, and neuropsychological outcomes to assess whether this novel cell-free regenerative technique can support surgical treatment and recovery in Moyamoya disease patients.
Want to stay updated on regenerative medicine trials? Get the free weekly newsletter here.