
Photoreceptor diseases, such as retinitis pigmentosa, cone-rod dystrophy, and Usher syndrome, are inherited retinal conditions that lead to the degeneration of light-sensitive cells in the eye. They often result in progressive vision loss and, ultimately, legal blindness. There are no approved treatments that restore lost photoreceptors, driving interest in cell-based regenerative therapies.


In this trial, participants will receive OpCT-001, which consists of photoreceptor precursor cells derived from human induced pluripotent stem cells (iPSCs). The therapy is delivered by subretinal injection directly into the affected area of the retina.
The study is divided into two parts:
- Phase 1 (dose escalation): Four planned dose levels of OpCT-001 will be tested across four cohorts. Each cohort will include approximately three to six participants, all of whom are legally blind. The dose escalation uses a standard 3+3 design to assess safety and tolerability.
- Phase 2a (dose expansion): Two selected dose levels, based on results from Phase 1, will be evaluated in up to 15 participants per cohort (total maximum: 30). Participants and investigators (except surgical teams) will be masked to the assigned dose.
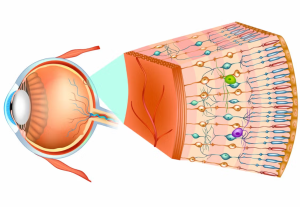
(Image Credit – BlueRockTherapeutics)
The primary objective is to assess the incidence and severity of ocular and non-ocular treatment-emergent adverse events (TEAEs) through 52 weeks after treatment. Secondary objectives include changes in the outer retinal layer thickness, as measured by spectral domain optical coherence tomography (SD-OCT), in the treated portion of the study eye over the same period.
The researchers note that OpCT-001 is a first-in-human application of iPSC-derived photoreceptor precursor cells for inherited retinal diseases. The expectation is that transplanted cells may engraft and provide functional benefit, though the main focus in early phases is on safety. According to the trial sponsors, OpCT-001 is being positioned as a potential regenerative approach for a group of disorders that are currently considered incurable. The therapy leverages advancements in iPSC technology, allowing for the scalable production of photoreceptor precursors with the goal of replacing or supporting lost retinal cells. The estimated primary completion date for the study is October 2029, with full study completion targeted for October 2030. Want to keep up on regenerative medicine trials? Get the weekly newsletter here.


















