Key findings
- A preclinical study reports that intranasal Muse cells reached the brain in a Parkinson’s mouse model and were linked to less neurodegeneration and improved motor behavior
- The authors propose that Muse cells cross the blood-brain barrier through S1P–S1PR2 (a signaling pathway that helps guide cells to injured tissue)
- Muse-treated mice showed markers associated with better preservation of dopamine-producing neurons
A new paper in the Journal of Translational Medicine reports that intranasally administered multilineage differentiating stress-enduring (Muse) cells reduced neurodegeneration and improved behavioral outcomes in a mouse model of Parkinson’s disease. The research was done at China’s Shandong First Medical University.
What are Muse cells?
They’re relatively “new” in regenerative medicine; here’s a quick introductory paper, summarized below.


In short, the authors of this paper claim that Muse cells exhibit both pluripotent-like characteristics (ability to differentiate into any or most cell types, as opposed to, for instance, mesenchymal stem cells, which have limited differentiation potential), and they appear to behave similarly to a macrophage. The researchers state, “they phagocytose damaged/apoptotic cells and differentiate into the same cell type as the phagocytosed cells,” meaning they may identify damaged cells, ingest them, and then differentiate into the same cell type as the ones ingested.
The researchers note several key hypothetical advantages over mesenchymal stem cells (MSCs): they may cross the blood-brain barrier and bypass the pulmonary first-pass effect, and they home to damaged sites at upwards of 15x the rate.
Are Muse cells a “Thing”?
They’re widely criticized, as is most of regenerative medicine, which is good. Dr. Paul Knoepfler, a stem cell biologist at UC Davis and the founder of iPSCell.com, said, “I am not so convinced that MUSE cells exist as a normal, easy to isolate population of stem cells that have substantial utility.” iPSCell ran a poll on whether MUSE cells exist at all, asking, “Are MUSE cells real adult pluripotent stem cells?” 44% of respondents said “probably not”.
Despite this, the Kardashians recently traveled to Mexico for Muse cells, posting about it on social media. It also appears that clinics are either offering or preparing to offer them in the United States soon (somehow?).
Difficulty in Treating Parkinson’s (BBB)
Parkinson’s, largely driven by the degeneration and death of dopaminergic neurons, is a challenge to treat with cellular therapies, partially because of the blood-brain barrier (BBB). You’ll see companies injecting directly into the brain to bypass this; for example, Bayer’s BlueRock Therapeutics is currently conducting an iPSC-derived neural cell trial in its exPDite-2 trial.
To evaluate the Muse cells’ ability to cross the BBB, they started with an in vitro BBB model, which showed that Muse cells migrated across it more effectively than non-Muse cells, especially under inflammatory conditions. They propose that the S1P–S1PR2 pathway (S1P is a signaling molecule released by injured or inflamed tissue) helps guide Muse cells to damaged brain regions.
The researchers also suggest that Parkinson’s-related inflammation may increase S1P levels, making the BBB more permeable and easier to cross.
How Could Muse Cells Help?
The authors propose that Muse cells home to damaged brain regions, reduce inflammation, and release neurotrophic factors (natural proteins that help brain cells survive, grow, and function), such as BDNF and GDNF.
Study design and Results
The researchers administered 1×10^6 Muse cells intranasally once per week for four weeks in a Parkinson’s mouse model (A53T).
They used non-Muse cells and PBS (saline) as controls, and to probe the homing mechanism theory, a subset of animals also received an S1PR2 antagonist (which blocks homing signals). Cell distribution was evaluated using fluorescent labeling and immunofluorescence in brain tissue.
The authors report that intranasally delivered Muse cells preferentially localized to the brain, while non-Muse cells largely accumulated in the lungs with little to no signal detected in the head region. Blocking S1PR2 reduced Muse cell brain signal, supporting a role for S1P–S1PR2 signaling in brain homing.
Compared with saline, Muse-treated animals showed signs of better dopamine-producing neuron survival, reduced inflammatory signals like TNF-α, and increased levels of those neurotrophic factors, BDNF and GDNF. These effects were diminished in the non-Muse and Muse + homing signal blocking groups.
Behaviorally, Muse-treated mice reportedly performed better:
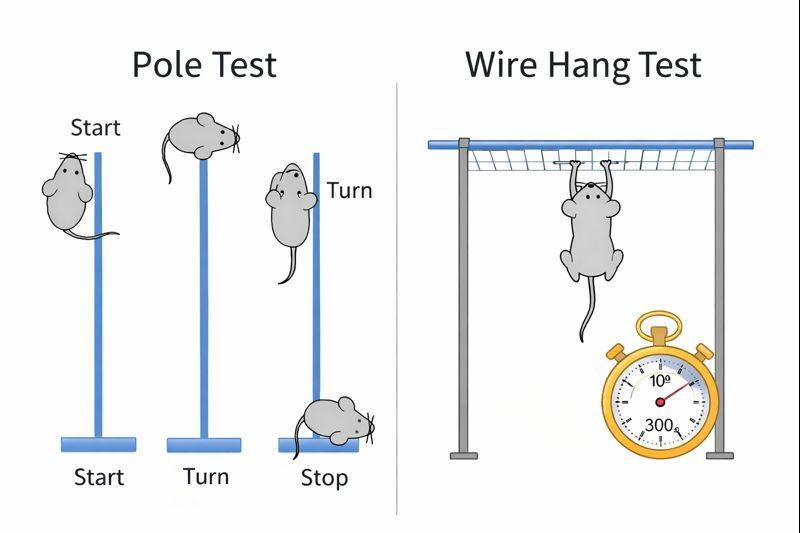
Pole test:
Climbing down was approximately 50-60% faster with Muse cells: Parkinson’s mice given PBS took about 13–14 seconds to climb down the pole, compared with roughly 5 seconds in healthy controls. Muse-treated mice improved to about 5–6 seconds, while non-Muse and Muse + homing signaling blocker group remained slower at approximately 8–11 seconds.
Turning was approximately 45–50% faster with Muse cells: PBS mice took about 7 seconds to turn, compared with about 2 seconds in controls. Muse treatment reduced turning time to roughly 3–4 seconds, while non-Muse and Muse + homing signaling blocker stayed closer to 5–6 seconds.
Suspension (wire hang) test:
Hanging time increased by approximately 70–80% with Muse cells: Healthy control mice hung for about 300 seconds, while saline-treated mice dropped to roughly 120 seconds. Muse-treated mice improved to approximately 210–220 seconds, whereas non-Muse and Muse + homing signaling blocker groups remained lower at around 160–180 seconds.
We have heard of a company preparing to launch Muse cells in the USA and potentially in Singapore in the near future. I’m uncertain how this would be allowed in the USA, as they may be allogeneic and more than minimally manipulated, and are relatively new (there are a handful of studies), but perhaps we’ll get more promising studies soon. It appears they are currently available in Eastern Europe and Japan.
The question is, are they ready?
Want to keep up on regenerative medicine? Get the weekly newsletter here.


















