Key findings
- Wharton’s jelly is a major source of mesenchymal stem cells and growth factors
- Wide variety of potential use cases
- Non-invasive harvest compared to other options
- Many in-human and preclinical trials are underway
Wharton’s jelly, the gelatinous tissue within the umbilical cord, is gaining attention as a promising, yet controversial, source of stem cells for regenerative medicine and tissue engineering.
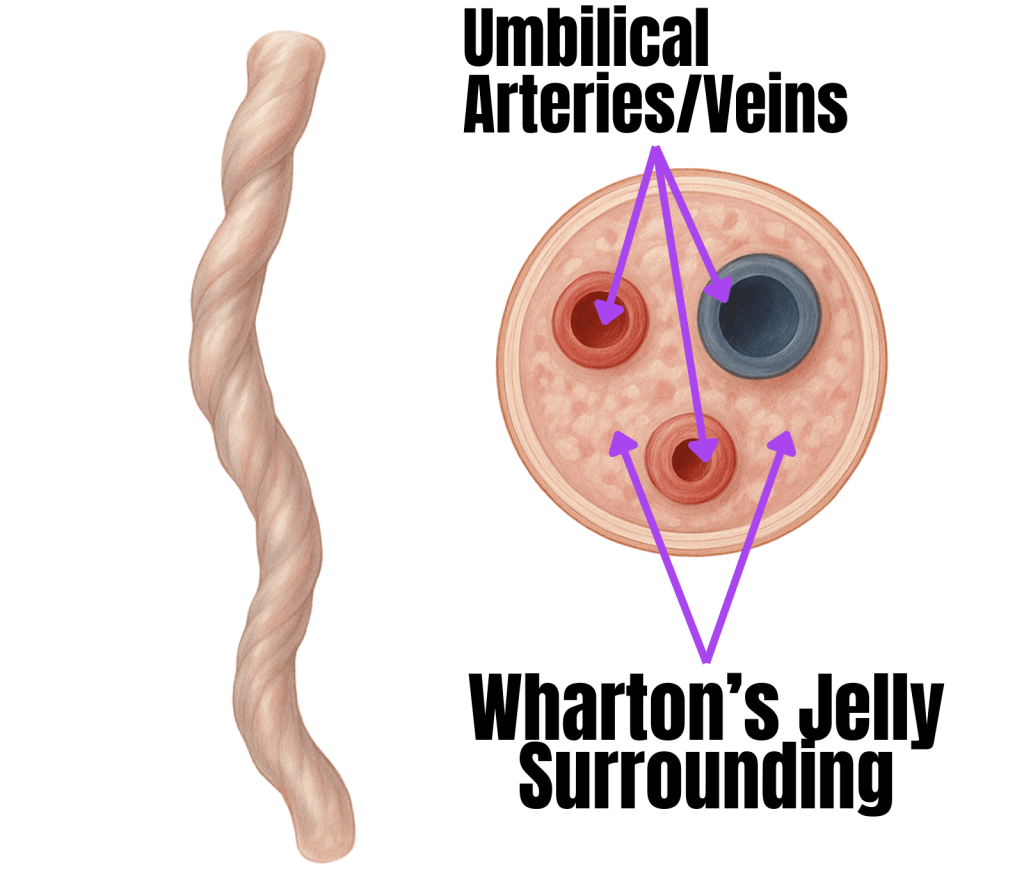
Wharton’s Jelly is a rich source of mesenchymal stem cells (WJ-MSCs) and growth factors, which may have a wide range of uses from orthopedics, autoimmune, and neurological conditions. There are a plethora of trials underway to investigate these conditions, while some clinics already offer the therapies ahead of FDA approval. Florida, for instance, recently enacted Senate Bill 1768, which allows physicians to administer WJ-MSCs under certain conditions, joining other states like Utah, Nevada, and others.


Unlike other sources of mesenchymal stem cells, such as bone marrow or adipose (fat), WJ-MSCs could have several benefits, most notably, non-invasive harvest and the potential to scale up dosages.
Bone marrow-derived MSCs, which have also shown promise, have a variety of limitations. MSCs account for only about .01-.001% of bone marrow. Oftentimes, patients are receiving very few MSCs from this method. Unfortunately, calculating the MSC content from a bone marrow aspiration requires expensive equipment that most clinics do not have on hand… meaning patients almost never know how many MSCs are administered. Of those MSCs, they may be prone to senescence and less capable of differentiation, especially in older patients.
Mothers, at least in the USA, often pay the hospital to incinerate the umbilical cord after birth, while this could be sold or donated to a tissue bank for other patients in need.
This makes an off-the-shelf birth-tissue MSC source, such as WJ-MSCs, a tempting therapy to investigate.
However, there are still many questions on cell viability, efficacy, and long-term clinical outcomes. In this new paper published in Biomedicine & Pharmacotherapy, researchers are calling Wharton’s Jelly a “Holy Grail” in tissue bioengineering and reconstructive medicine.
Unlike many other journals where WJ research is published, this journal has an impact factor of approximately 7.5. Here’s what the researchers said, with some additional context added by Regen Report.
Regenerative potential for many conditions
The authors highlighted that WJ-MSCs demonstrate robust proliferation and the ability to differentiate into multiple cell types, which could be valuable for a variety of medical needs ranging from musculoskeletal to vascular, neurodegenerative, and even autoimmune conditions.
Spinal cord injury – WJ-MSCs have shown some promise, albeit limited in this study. In this new Spanish trial, researchers appear to be upping the dosages and adding in transcutaneous stimulation + neurorehab to see if they can improve outcomes.
Orthopedics – In my opinion, this appears to be the most common application of WJ-MSCs, often touted by Joe Rogan and others on the internet. In this study, researchers found significant MRI improvements in knee osteoarthritis using expanded WJ-MSCs, while this study cited by the paper shows WJ-MSCs may support chondrogenesis (the creation of cartilage cells).
Autoimmune – The researchers noted that WJ-MSCs may possess immunomodulatory properties, which may reduce inflammation and tissue repair. There is one single MSC therapy approved by the FDA, it’s called Ryoncil, a bone marrow-derived MSC therapy that modulates the immune system for juvenile graft-vs-host-disease. Interestingly, in the FDA’s summary of action letter, it was noted that the “mechanism of action of RYONCIL is unclear”, yet it still gained approval based on its efficacy.
Neurodegenerative diseases – Alzheimer’s, Parkinson’s, ALS, and many other neurodegenerative conditions are finding some level of potential with WJ-MSCs, albeit sometimes limited. This ALS study reported 31% of patients experienced a decrease in progression rate. It’s also been studied that WJ-MSCs may differentiate into Schwann-like cells, promoting recovery of nerves, which may be one mechanism of action for WJ-MSCs.
Brain Recovery – Additional research is being conducted for strokes and traumatic brain injury, which has shown some promise in preclinical research.
Tissue Grafts and Biomaterials
Relatively new, but heating up in the regenerative space, some researchers are finding early success using WJ-MSCs as seeds for tissue grafts, such as nerve grafts.
Wharton’s Jelly, even decellularized versions, have been used in many vascular repair studies as well, which may promote cell migration and endothelial growth.
Some researchers are attempting to mix and match WJ-MSCs with other biomaterials for better efficacy. For instance, a 3D printed scaffold and hydrogel, which may create an environment favoring MSC proliferation and differentiation into osteocytes (bone cells), or hyaluronic acid, which may act as a scaffold for better spinal cord repair.
Want to keep up on regenerative medicine research and news? Get the weekly newsletter here.