Interview: Veterinary Stem Cells – Gallant’s Quest for the First FDA-Approved MSC Therapy
Gallant is betting the future of veterinary regenerative medicine lies in scalable, well-characterized cell therapies. By sourcing mesenchymal stem cells from uterine tissue collected during routine spays, it has built an off-the-shelf platform with consistent, high-yield cells and FDA-aligned manufacturing, with a potential first FDA approval on the horizon.

In this article:
- Intro to uterine-derived mesenchymal stem cells
- How these cells compare to other sources
- The clinical and manufacturing advantages
- An inside look at their clinical trials, including a potential historic FDA approval
- Current and future state of the veterinary stem cell industry
Regen Report: In 2019, a vet biotech company named Gallant appeared on Shark Tank, inking a $500K deal to develop stem cell therapies for pets.
Years later, they landed another $15M in Series A financing and have used this capital to raise the scientific bar not only for veterinary medicine but also for regenerative medicine as a whole, developing novel techniques to harvest and apply uterine-derived mesenchymal stem cell (MSC) therapies.
Feline Stomatitis is among the leading indications, and this program is poised to make history as the first FDA-approved off-the-shelf veterinary MSC treatment, with conditional approval anticipated in 2026.
We sat down with veterinary neurologist, Dr. Rebecca Windsor, DVM, DACVIM, Director of Veterinary Affairs at Gallant, who outlined the cells, trials, results, and the company’s hypotheses so far.
Enjoy the interview!

Can you give us a quick history of Gallant?
Rebecca: In 2017, our late founder, Aaron Hirschhorn, found significant relief for his back pain with MSC therapy, leading him to research MSCs for his geriatric dog suffering from arthritis.
Like in humans, we’ve had same-day autologous (sourced from the patient themselves) MSC procedures for a couple of decades, but after consulting a variety of scientists, he concluded that maybe the cell source shouldn’t be from an old, unhealthy patient, but rather allogeneic cells (sourced from a donor) from young, healthy ones.
This led to the founding of Gallant in 2018, with a two-fold mission:
- Banking autologous MSCs while pets are young
- Developing off-the-shelf MSC therapies for the broader industry
MSCs can be harvested from a variety of tissues, including adipose (fat), bone marrow, and perinatal tissues such as the umbilical cord, placenta, and amniotic fluid.
However, about 20 years ago, scientists discovered that the uterus is also rich in MSCs, progenitors, uterine stem cells, and other regenerative cells. Gallant sources our off-the-shelf MSC therapies from uterine tissue collected during routine spays of healthy donors, which will be the focus of this interview.
Uterine Derived MSCs
Rebecca: It makes sense that the uterus is rich in regenerative cells because its natural function is to support the growth of a new life, and it sheds and regenerates itself over hundreds of reproductive cycles before menopause.

In the US, we spay/neuter our pets, then discard that young, regenerative tissue. Rather than wasting it, our team developed a platform that enables us to harvest MSCs from uterine tissue, and we’ve identified several advantages over currently available MSC therapies.
Advantages over Today’s Stem Cell Therapies
Rebecca: Let’s first introduce today’s methods of autologous MSC therapy, often harvested and processed in-house or sent to a lab before administration, and compare them to our allogeneic off-the-shelf MSC therapies.
The two most common methods today are:
- Bone marrow concentrate (BMAC) – Bone marrow is aspirated, typically from the iliac crest, centrifuged, and the buffy coat layer, which contains MSCs, hematopoietic (blood) stem cells, growth factors, and platelets, is injected.
- Stromal vascular fraction (SVF) – Adipose tissue is surgically removed and then enzymatically digested, yielding a heterogeneous pellet of cells, including MSCs, which is then injected.
Although there are certainly reports of improvement, these methods are largely unregulated, and there is enormous variability in what is actually administered. Both methods are also invasive procedures that yield very low MSC counts.
Compared to our off-the-shelf allogeneic therapies, we’ve found several advantages for patients:
1 – FDA Approval
Rebecca: BMAC and SVF forego FDA approval, allowing them to reach the clinic without trials, creating a mystery around what’s injected and how efficacious it is (more on that later).
However, allogeneic cell therapies typically require FDA approval, meaning when they reach the clinic, you can trust that the safety, efficacy, and manufacturing consistency have been well demonstrated. We see this as an opportunity to set the bar and build trust in the industry.
2 – Control Over Cell Quality and Quantity
Rebecca: According to the peer-reviewed literature, bone marrow’s MSC content can vary widely, ranging from 0.001%-0.01%. Unfortunately, clinics typically lack the equipment or expertise to count MSCs, so the true MSC count remains unknown. Without culture and expansion (the process of duplicating stem cells in a lab, which increases their numbers), the final population is likely about 1000 MSCs in 1 mL of bone marrow.
Similarly, the literature on canine adipose states that about 0.1% of the cell population is MSCs, which is 10-100x that of bone marrow. Although SVF also contains a number of anti-inflammatory factors, it’s a heterogeneous mix with a low percentage of MSCs. Like BMAC, there’s rarely any cell characterization or quality control; it’s simply harvested, processed, and injected. In animals, collecting fat requires sedated surgery, and MSC density varies widely depending on patient age, size, health, and harvest site.
Regen Report: Some companies question whether these should even be considered “stem cell therapy”, given the small number of MSCs.
3 – Volume
Rebecca: Another downside of BMAC and SVF is the limited and unpredictable amount of bone marrow and fat to draw from, especially in small animals.
Not only is uterine tissue a rich source of MSCs (our scientists estimate 3-7% of our uterine cell population is MSCs), but it’s also more homogenous, and the tissue yield is more predictable.
(Pullquote) – To give a sense of our yield, after culture and expansion, one spay produces over 30 million doses of well-characterized product, each containing 20 million MSCs.
This means one spay can help millions of patients.
Ethical, sustainable use and scalability are among the main reasons we chose the uterine-derived MSC platform.
4 – Harvesting and Accessibility
Rebecca: Today’s MSC therapies shift the burden of tissue collection to the veterinarian and the patient. Not only are they painful and invasive, but most vets aren’t comfortable performing them. Outside of oncology, most vets only perform a few bone marrow aspirations during school.
Using an off-the-shelf product can eliminate this burden.
5 – Younger Cells
Rebecca: Studies have shown that as patients age, their MSCs proliferate, migrate, and immunomodulate less well. This is why most platforms are shifting toward cells from young, healthy donors, although many are not undergoing FDA approval to ensure consistent safety, efficacy, or product composition.
Novel Variables
Rebecca: There are also new challenges to address when going off-the-shelf:
1 – Pathogenicity
Rebecca: “Healthy donor” is a relative term, and its definition needs to be tightly controlled. In some cases, “healthy” animals may still be infected with pathogens that affect stem cell numbers or quality, or, even worse, transmit infection to a patient.
We source uterine tissue from a small number of dogs and cats raised in a specific-pathogen-free colony. These animals are thoroughly screened for infectious agents and meet FDA systemic health standards. We empower veterinarians by ensuring that when they buy our allogeneic products, they know exactly where they were sourced and how the donors were screened.
2 – Immunogenicity
Rebecca: MSCs are generally considered “immune-evasive” because they express low levels of MHC class I receptors, allowing them to evade detection by the host immune system.
They also appear to promote an immune-tolerant environment. In humans, a low percentage (11.5%) of patients form antibodies to donor MSCs, but this does not appear to affect the safety or efficacy.
Many studies exploring MSC immunogenicity and host rejection are performed in vitro, where the concentrations of MSCs and host cells are much higher than in the body, likely falsely elevating antigen exposure.
These in-vitro studies also lack the anti-inflammatory/immune-tolerant environment that MSCs create in vivo. These factors may falsely amplify a potential immune response.
That said, we’re still learning, and continuing large, controlled, in vivo, real-world studies with long follow-up times is essential. From a safety standpoint, we have treated hundreds of patients with allogeneic cells. Adverse events are mild, typically self-limiting, and don’t appear to increase with repeat dosing. We also haven’t seen any long-term adverse events associated with immunogenicity.
3 – Manufacturing and Logistics
Rebecca: Part of the FDA approval process is validating our CMC (Chemistry, Manufacturing, and Controls), which is extensive and in many ways a greater accomplishment than demonstrating safety and efficacy. Once validated, it means vets and pet owners can trust the product will be consistent across doses and batches.
After they’re manufactured, the cells are stored at ultra-low temperatures before being shipped for on-demand use. We store the cells in -80°C freezers and can ship them on dry ice or store them in small in-hospital freezers.
One nice thing about using injectable therapeutics is that control over ordering, dosing, and monitoring is placed back in the hands of the veterinarian, rather than having owners order drugs online with less clinician oversight.
4 – Scientific Evidence
Rebecca: Autologous cell therapies have been used in veterinary medicine for the past 25 years, particularly for osteoarthritis in dogs and horses, so naturally the scientific evidence/literature base is larger.
Allogeneic therapies have been increasingly used over the past 10 years, and for many diseases (feline chronic gingivostomatitis, chronic kidney disease, atopic dermatitis, chronic enteropathy), they are now the standard of care. The low patient numbers and large variability in stem cell source, dose, frequency, and administration route make it difficult to compare studies, but many groups, including ours, are leading the charge to pursue large, randomized, blinded, controlled clinical trials to better assess.
Are Uterine-Derived MSCs “Better”?
Rebecca: It’s rarely as simple as one tissue source being “better” than another. We believe it’s most important to use well-characterized products sourced from young, healthy donors.
That being said, there are certainly differences in cytokine profiles (signaling proteins secreted by cells) between sources, which could influence their immunomodulatory potential. Different sources also have distinct differentiation profiles (what type of cells they are likely to become), and some may proliferate better than others.
And even within a single tissue source, you can see meaningful differences from cell line to cell line. This is where knowing the underlying pathology associated with the targeted condition is so helpful. For example, some uterine tissue has cytokine patterns that make that cell line well-suited to treat stomatitis, while others may show profiles well-suited for osteoarthritis or atopic dermatitis.
To really compare tissue sources, we would need to do a head-to-head study using the same patient population, dosing, clinical outcome measures, and, in some cases, pathology and biomarkers to understand what’s happening at the molecular level, which is difficult to achieve.
I’ve treated patients with both adipose- and uterine-derived MSCs using the same dose and protocol, and both have worked well. The most important takeaway is that it isn’t about whether you’re using cells derived from bone marrow, fat vs uterine tissue, it’s about the characteristics of that specific cell population from that particular lab, which is again why FDA approval is so crucial.
Which Conditions Are You Treating, Why, and How Is That Progressing?

Rebecca: If we had unlimited resources, there are so many conditions that may benefit, but we must choose where to focus. We decided to find a few conditions with limited available treatments, share underlying pathology, and that can be treated similarly.
This led us to:
- Gingivostomatitis (stomatitis)
- Osteoarthritis (OA)
- Chronic kidney disease (CKD)
- Atopic dermatitis
Which are all in varying stages of trials as we speak. Even though these conditions appear clinically distinct, they are likely rooted in systemic immune dysregulation that may manifest in the joints, skin, or mouth, which aligns with one proven mechanism of IV MSC therapy: immunomodulation. As of January 2026, there is one single FDA-approved MSC therapy in humans, Ryoncil, administered IV for pediatric refractory graft-vs-host disease. It’s proven to modulate the immune system.
Administration and Dosage:
Rebecca: We are currently using 20 million cells IV for most indications, and in the long term, we’ll evaluate whether lower dosages are also effective.
We should point out that there’s been concern that IV MSCs become trapped in the lungs due to pulmonary first-pass, and that these cells may not reach target tissues. This is based on a 25-year-old study in healthy rats given IV MSCs, but the rats were healthy, and likely the MSCs didn’t have anywhere to home to.
(Pulmonary first pass chart, with micrometer scale if possible)
Since then, several studies in humans and animals have shown that MSCs can home to target tissues, such as the mouth in cats with stomatitis, joints in dogs with osteoarthritis, among others.
MSCs also appear to predominantly act through immunomodulation rather than tissue engraftment, and much of their beneficial effects occur in the bloodstream, where they secrete cytokines, growth factors, and extracellular vesicles that modulate the systemic immune system, and in the lungs, where they convert the macrophage population from a pro-inflammatory to an anti-inflammatory type. These macrophages can also circulate systemically.
Gingivostomatitis
Rebecca: Gingivostomatitis (stomatitis) is a brutal disease affecting about 10% of the feline population. Cats develop ulcers and, in some cases, mass lesions in the back of the mouth, sometimes large enough to block the airway. Almost all have esophageal inflammation and respiratory involvement, and about 30-50% of these patients are euthanized.
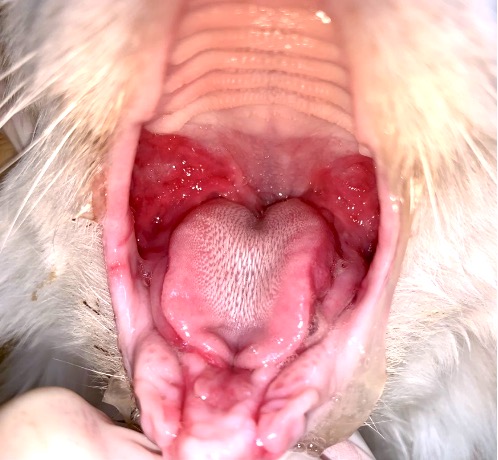
The etiology is complicated; we think it’s an aberrant immune response, at least in part due to plaque bacteria. We also think it may involve an immune response to a viral infection, as about 75-80% of these cats are viral positive, usually for feline calicivirus.
The standard treatment is tooth extraction with medical management using immunosuppressants, but about a third of them remain refractory despite aggressive therapy. There’s a strong body of research from UC Davis using adipose-derived IV MSCs for this condition that sets the stage for Gallant’s exploration of uterine-derived IV MSCs for refractory FCGS.
Can You Talk About the Results?
Rebecca: In short, we expect about ⅔ of cats are likely to improve.
One of the challenges in stem cell studies is the inconsistency in cell source, dosage, administration route, and frequency. It often ends up as a series of anecdotes, making efficacy difficult to quantify and compare.
However, having two institutions use similar protocols and find consistent outcomes helps confirm and support the validity of the data. We measure outcomes similarly to UC Davis, using a combination of owner-reported assessments, client-specific outcome measures, quality-of-life improvement, and veterinarian-assessed global oral lesion score.
In September 2025, we published short-term data demonstrating:
- By day 90, 75% showed improved quality of life
- By day 365, 66% sustained this improvement
- Lesions heal more slowly, but about 50% showed significant improvement by day 365, and many without significant lesion healing still appear to improve in quality of life
- Finally, almost 25% of cats return to normal, which is amazing given the refractory nature and high euthanasia rate of this population
This seems to align with the UC Davis studies, which reported a roughly 65% long-term response rate.
Because the condition is so serious, widespread, and lacks therapeutic options, it’s an excellent candidate for conditional FDA approval. If all goes well, we’re hoping to have the first FDA-approved stem cell therapy in veterinary medicine in the summer of 2026.
What’s the Mechanism of Action?
Rebecca: This condition is likely rooted, at least partially, in T cell dysregulation. Our evidence shows that MSCs help reduce T cell expansion, reset helper T cell and cytokine patterns towards a more balanced immune profile, and impact viral activity by enhancing T cell antiviral function and slowing viral replication.
This seems to align with evidence that immune dysregulation is at the core of this condition and that MSCs help reset this imbalance. This also tracks with Ryoncil’s immunomodulatory mechanism of action.
Osteoarthritis (OA)
Rebecca: OA is among the most studied indications for stem cell therapy in both humans and animals. About 80-90% of geriatric cats and 40% of dogs over age 4 have some form. Our CEO, Dr. Linda Black, DVM, PhD, is one of the pioneers of stem cell therapy in veterinary medicine and authored some of the earliest RCTs using intra-articular MSCs for dogs with OA.

OA was historically thought to be an age-related “wear and tear” disease, but data increasingly support that underlying inflammation and immune dysregulation are big factors. This suggests we should explore tools to diagnose and treat OA earlier in the disease course rather than waiting until joints are severely damaged.
Intra-articular injection is common; however, MSCs have been shown to home to inflammation sites, and several studies report good success with IV administration. If that’s the case, IV would bring several advantages, including:
- Better accessibility: Intra-articular injections require specialists, such as rehab or surgical vets, with advanced imaging equipment, while IVs are easy and routine for any vet or even technician.
- One injection, multiple joints: OA often affects multiple joints. IV administration could target many joints simultaneously by homing to sites of inflammation in multiple areas. This lowers the cost because you don’t need multiple injections.
- Less sedation and recovery time: Intra-articular injection requires sedation, which incurs additional cost, sedative risk, and recovery time. IV does not.
We are completing enrollment in our pilot trial soon and hope to make decisions on the next phases in the coming months.
Feline Chronic Kidney Disease (CKD)
Rebecca: Feline CKD was a clear target because it’s a common, debilitating, and leading cause of death in cats. When CKD damages the nephron, it’s typically irreversible and can be progressive.
The thinking has evolved from CKD being purely degenerative to now understanding the inflammatory triggers that can drive and sustain the disease. If we can target that inflammation, could we potentially improve outcomes for a disease that’s historically been considered irreversible?
One challenge in finding out is accurately measuring renal function. We have relatively crude markers, such as creatinine, but more functional measures, such as GFR (glomerular filtration rate), are technically complex and not practical in a clinical setting. We have completed enrollment for this phase of the CKD trial and will be deciding over the coming months what the next steps are.
Atopic Dermatitis
Rebecca: Atopic dermatitis is another compelling indication because it is strongly rooted in immune dysregulation, and there is a substantial foundation of both animal and human model data to build on.
Current treatments are effective at reducing pruritus (itching) and inflammation, but they rarely address the underlying disease. As a result, many patients remain chronically medicated, which can be expensive, burdensome, and difficult to sustain.
Atopic dermatitis is a very complex disease. There are multiple triggers, and secondary bacterial and yeast overgrowth commonly exacerbate symptoms. Immune signaling also changes over time, with a predominantly TH2-mediated response in acute disease and a shift toward a more TH1-driven profile in chronic phases. That complexity is part of what makes stem cells interesting in this context; they appear to “read” the environment and modulate their activity in response to those signals.
There have been promising phase 1 and 2 human studies, and several canine studies have demonstrated improvements in pruritus, lesion scores, and, in some cases, epidermal thickness after MSC treatment, suggesting skin repair. The challenge in comparing these studies is the wide variability in dosing and administration protocols.
The trial is a bit behind the others because we opened it later and, initially, like other similar trials, required dogs to taper medications prior to enrollment.
This is a tough ask because allergy symptoms will undoubtedly increase for most dogs, half of whom will receive a placebo. We’ve since shifted to a more realistic scenario in which dogs remain on stable doses of their drugs, and enrollment is finishing for the current phase. We’ll soon decide on the next steps.
Almost 10 Years In, What’s Surprised You the Most Along the Way?
Rebecca: One that sticks out is just how enthusiastic vets and pet owners are to participate. It’s clear that people need a new option that addresses the root cause of disease rather than just symptom management with long-term medication.
It’s also been great to see how collaborative the regenerative medicine community is, particularly among those committed to developing therapies the right way.
How do you see the current stem cell industry changing?
Rebecca: Historically, the stem cell industry has had a “wild west” feeling. Given the limited oversight of today’s autologous therapies, most are selling products based on marketing rather than science.
It’s really hard to develop a safe, proven product, let alone manufacture it at scale while retaining quality, most companies don’t even try…
Despite that, many allogeneic products make it to the clinic without going through proper channels; sometimes they’re shut down or receive FDA warning letters, but reinforcement seems slower on the veterinary side.
It’s common for these platforms to rely on anecdotal “evidence,” such as a photo of a dog who “did great,” as an effective promotional tactic. All too often, these therapies provide little to no clinical improvement, which causes patients and clinicians to dismiss stem cell therapy as a whole. Our job is to teach people how allogeneic off-the-shelf MSC therapies developed in a GMP lab may differ.
We also see many people, even vets, still associate stem cells with embryo sourcing, tumor risk, and immune reactions, so my job as Director of Veterinary Affairs is often to relay how our MSCs are different and the honest, scientific reality.
As for the future, it’s such an exciting time for regenerative medicine. So much is coming out so quickly, it’s honestly hard for me to keep up with the literature! Research into other cell-based therapies, including stem cell-derived extracellular vesicles and induced pluripotent stem cells (iPSCs) are helping promote awareness and drive interest. Although both have increased risk profiles and logistical hurdles that must be overcome before they can be clinically practical.
As for Gallant’s future, we look forward not only to introducing new drugs but to launching a new category of care. We’re committed to taking all necessary steps to ensure our off-the-shelf MSC therapies are sustainable in the long term, and we’re excited to bring therapies that may treat disease at its source rather than managing symptoms.
Regen Report: Special thanks to Dr. Rebecca Windsor for the interview! Hopefully, we hear a historic announcement this year. Wish them luck!