Aspen Neuroscience Secures $115 Million Funding for Parkinson’s Stem Cell Therapy
The new funding round will help advance their iPSC-derived dopaminergic neuronal precursor cell trials, manufacturing, and continued development of other iPSC therapies.

Aspen Neuroscience has closed a $115 million Series C financing round to continue development of its lead stem cell candidate, ANPD001, for moderate-to-advanced Parkinson’s disease.
ANPDD001 is an autologous iPSC-derived dopaminergic neuronal precursor cell (DANPC) therapy, and it’s currently in Phase 1/2a trials. They mention they’ve developed a proprietary delivery system that combines metered-dosing syringe technology with MRI guidance, enabling sub-millimeter accuracy during transplantation.
With this latest round, including an $8 million grant from the California Institute for Regenerative Medicine (CIRM), Aspen’s total capital raised now exceeds $340 million. They’ll also gain a new board member, Cindy Perettie, Executive VP from one of the funding partners, Kite.
Get Regen Report in your inbox
Weekly updates on regenerative medicine news, trials, and regulatory moves.
Plans for Funding:
- Support ongoing Parkinson’s trials
- Expand manufacturing capabilities
- Advance additional autologous iPSC-derived therapies targeting other neurological conditions
Here is the pipeline according to their website:
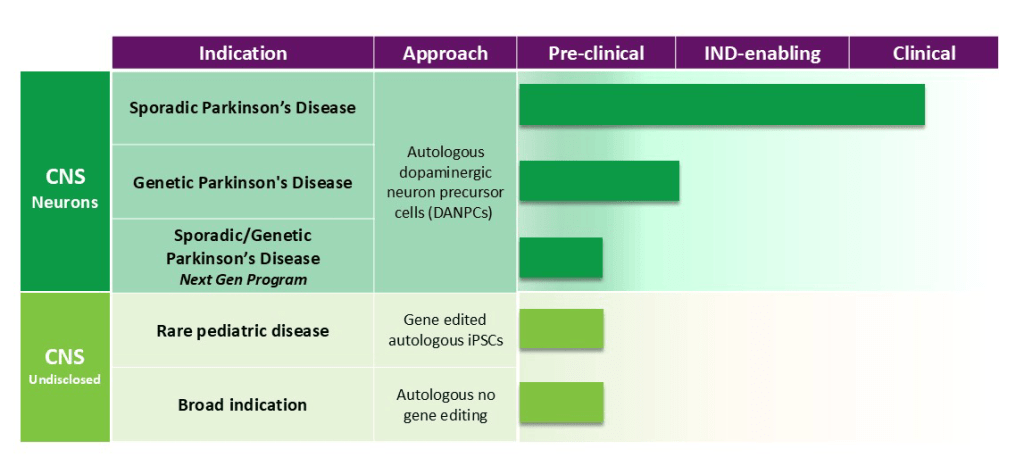
Aspen also mentioned that it has developed a proprietary manufacturing platform that uses machine learning combined with advanced genomics to produce high-quality personalised cells for each patient, and they’re aiming to improve consistency at a larger scale.
Recent Progress
- Cohort 3 dosing has started in the ASPIRO Phase 1/2a trial, which now uses a commercial-ready formulation of ANPD001. According to preclinical studies, this formulation is comparable to those used in earlier cohorts.
- This new formulation is cryopreserved, ready-to-dose, and will hopefully simplify workflows for hospital labs.
- Previously reported six-month data from earlier cohorts showed strong safety/tolerability, along with both clinician- and patient-reported improvements, all without immunosuppressive drugs (as opposed to allogeneic cells, which may cause an immune response)
For further details, visit their website: www.aspenneuroscience.com.
Want to keep up on regenerative medicine? Get the weekly newsletter here.








